If you search segregation in google, first few hits will be about racial segregation; strictly speaking I am not going to talk about it. It’s the segregation in materials world is my cup of tea. While working with oxide ceramics defects are something that never left me and now I say I love defects! There are classic effects you get to see due to presence of point and line defects in the materials, whether its semiconductor or any structural applications. Segregation is a results caused by point defects. It has been exploited in many ways. Optoelectronic, Transistor, structural ceramics, there are numerous occasions when segregation is the key. In the ionic solids, the Schottky defects causes ions to go to the interfaces, primarily they require a lattice discontinuity, so this place may be a surface, a grain boundary or any interface. Thus there is a local charge builds up there and a potential difference between the bulk and the surface is generated. The magnitude depends on the defect concentration, temperature. Like colloids there exists an isoelectric point where surface charge becomes zero and then with an increase in temperature or defect concentration it reverses. Segregation is a subject of interest of the structural community as this process retards grain growth. The space charge which forms at the boundary actually pins the grain boundary mobility. Sounds interesting? It has more effects; when you reduce the grain size that is in the nanometer range lot of material properties in oxide ceramics is controlled by segregation, hope you didn’t believe it! Segregation captures a huge part in grain boundary engineering. Si3N4 brittle in nature but it is toughened by adding some dopants, but how it helps was not known till very recently. Last year a paper in nature showed experimentally evidences of La segregation in La doped Si3N4, La actually segregates to the boundary and helps in forming elongated grains which in turn toughens the material. Following figure is actually taken from the paper.
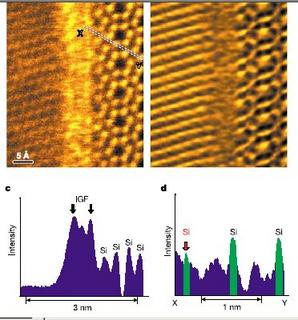
N. Shibata et al. Nature Vol. 428, pp730-33, 2004.
This is an STEM micrograph of a grain boundary in La doped Si3N4, bright fringe in the middle correspond to the amorphous film and the brighter spots in that are actually La. Segregation in YTZ is in discussion for a while and now it is said that nanocrystalline YTZ are more creep resistant that the submicron grain one because of Y+3 segregation. It is known that Y+3 play a role there but it is not understood which role it plays, so lots of questions. Sometimes segregation is detrimental, YTZ although has potential use as bone replacement, can not have an immediate use as Y segregation causes brittleness of the material. There are lots of interesting stuffs in this field and I am happy as I will have jobs in hand till the questions are answered. I really like defects.
1 comment:
Santa,
I think it is Barsoum but I might be wrong. Anyway, there are two quotes. One is called the "Dance of the solids", I suppose. Googling might get it for you - It talks about defects. The other one says "Crystals are like people. Only defects make them interesting".
So, I guess you are not alone in loving defects. Anyway, fundoo piece of writing. And before I sign off, here is Shakespeare (quoted in Westbrook - Intermetallics): "What is the cause of this defect, or effect, for this effect defective comes from a cause". I hope I got it right.
Guru
Post a Comment